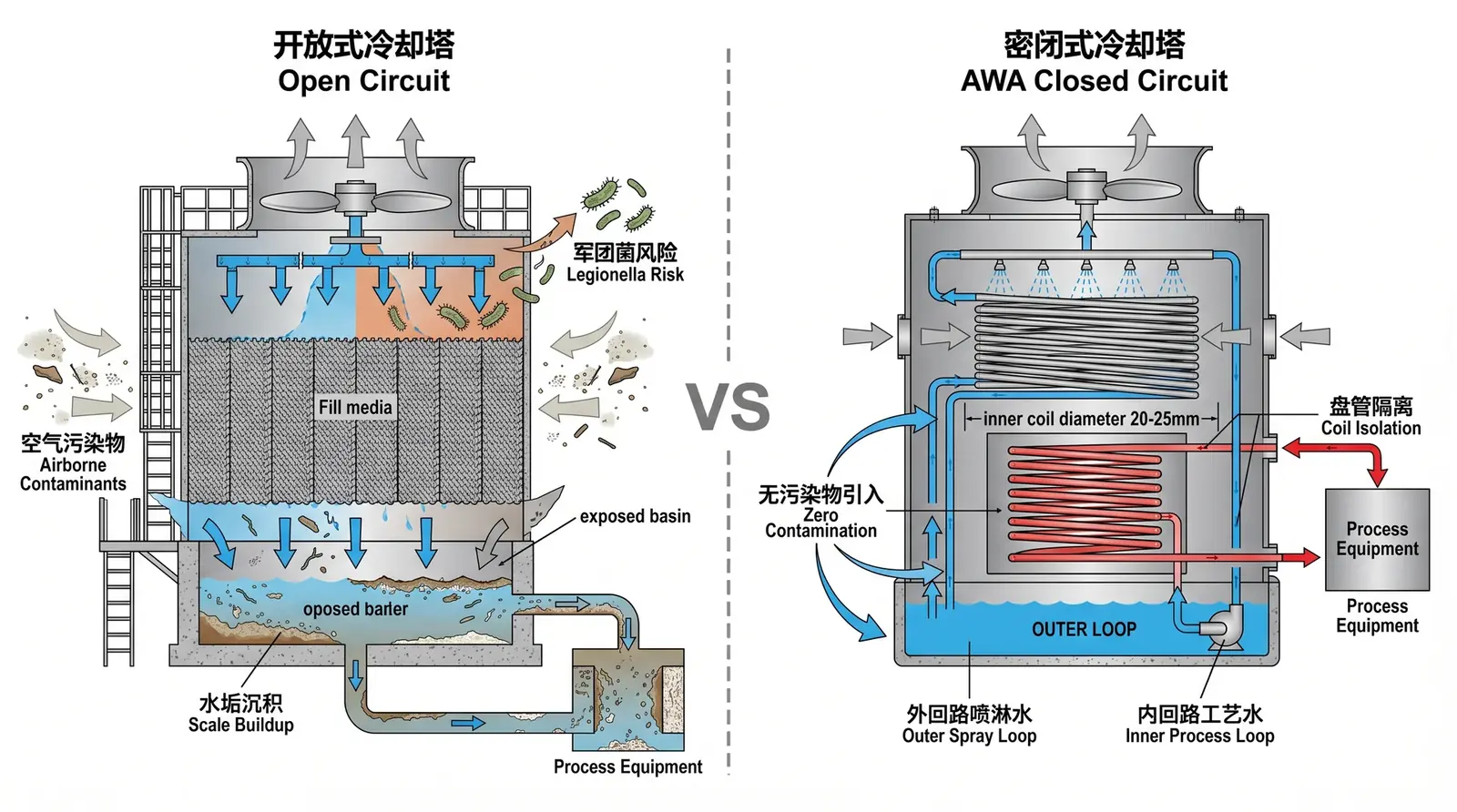
Figure 1: An open tower (left) introduces contaminants through air scrubbing and evaporative concentration. The AWA closed circuit (right) cuts off all contamination paths through physical isolation.
Problem Definition: The Core GMP Requirement for Cooling Water
The core GMP requirement for cooling water systems in pharmaceutical plants can be summarized in one sentence: the cooling water system must not become a pathway for microorganisms or chemical contaminants to enter the pharmaceutical production environment.
This requirement is clearly reflected in the regulatory framework applicable in Vietnam. Article 3.36 of TCVN 10405:2015, Vietnam's GMP standard, requires that "utility systems, including cooling water, should be validated to demonstrate that they will not adversely affect product quality." WHO GMP TRS 986 Annex 2, Article 14.1, requires that "cooling water systems should be regularly monitored for microbial load."
Physical Principle: The Scrubber Effect of an Open Tower
The fill section of an open cooling tower is not only a heat rejection component, but also an efficient gas-liquid contactor. It has a significant scrubbing effect on the air passing through it. In a pharmaceutical environment, this physical characteristic creates three compliance risks.
How microorganisms captured by the fill multiply exponentially in circulating water
Microorganisms in the air, including Legionella, Pseudomonas aeruginosa, and fungal spores, are captured by the circulating water in the fill and multiply rapidly in warm water at 25–35°C. Under the most favorable temperature, around 35°C, the doubling time of Legionella is about 4–6 hours. In open-tower circulating water without effective disinfection, Legionella concentrations may rise within a few days to levels more than 10 times above the WHO guideline value of <10⁴ CFU/L.
How organic solvent vapors and environmental exhaust gases enter water through the scrubber effect
Organic solvent vapors discharged from pharmaceutical cleanrooms, such as ethanol and acetone, disinfectant aerosols, and SO₂ and NOₓ from the surrounding environment can all enter the circulating water through the scrubber effect of an open tower. After these substances concentrate in the circulating water, they may enter process cooling water through heat exchanger leakage, creating cross-contamination risk.
How drift aerosols containing contaminants threaten the fresh air system of cleanrooms
During operation, an open tower produces drift aerosols containing droplets of circulating water. These droplets are typically 1–100 μm in diameter, can remain suspended in air for several hours, and can travel several hundred meters. If the drift contains microorganisms or chemical contaminants, it can directly enter the fresh air system of a cleanroom.
COOLTEK Solution: The Compliance Path of the AWA Closed Circuit
The AWA closed-circuit cooling tower meets GMP compliance requirements through the following mechanisms:
- Physical isolation: Process water circulates inside sealed coils. The coil wall forms a physical barrier between the process water and external air. Microorganisms and chemical pollutants in the air cannot enter the inner circuit.
- Drift control: AWA is equipped with a high-efficiency drift eliminator, with drift loss below 0.001% in accordance with CTI standards, greatly reducing aerosol emissions that contain spray water droplets.
- Verifiability of the inner circuit: Water quality parameters in the closed inner circuit, including conductivity, pH, microbial load, and corrosion and scale inhibitor concentration, can be continuously recorded through online monitoring instruments, forming the complete validation data chain required by GMP, including IQ/OQ/PQ.
Standards Verification: Specific Requirements of TCVN 10405 and WHO GMP
| Standard Clause | Requirement | Compliance Difficulty for Open Towers | Compliance Path for AWA Closed-Circuit Tower |
|---|---|---|---|
| TCVN 10405 Article 3.36 | Utility systems must not adversely affect product quality | Continuous monitoring and chemical treatment are required, resulting in high validation cost | Physical isolation; stable inner-circuit water quality; validation data is easier to obtain |
| WHO GMP TRS 986 Article 14.1 | Regular monitoring of microbial load in cooling water | Microbial load in open towers fluctuates widely, requiring high monitoring frequency | Microbial load in the closed circuit is stable, allowing monitoring frequency to be reduced |
| ISO 11731 (Legionella testing) | Legionella in cooling water <10⁴ CFU/L | Continuous chlorination or other disinfection treatment is required | The closed circuit has no favorable conditions for Legionella growth and usually requires no dedicated disinfection |
Extended Questions
- What documents and test items are required for AWA closed-circuit inner-loop water quality validation, including IQ/OQ/PQ?
- If a plant is already using an open tower, what are the approximate downtime and retrofit costs for conversion to a closed-circuit tower?
- Does the drift eliminator of a closed-circuit tower need regular replacement? What is the replacement interval?